You're likely already infected with a brain-eating virus you've never heard of
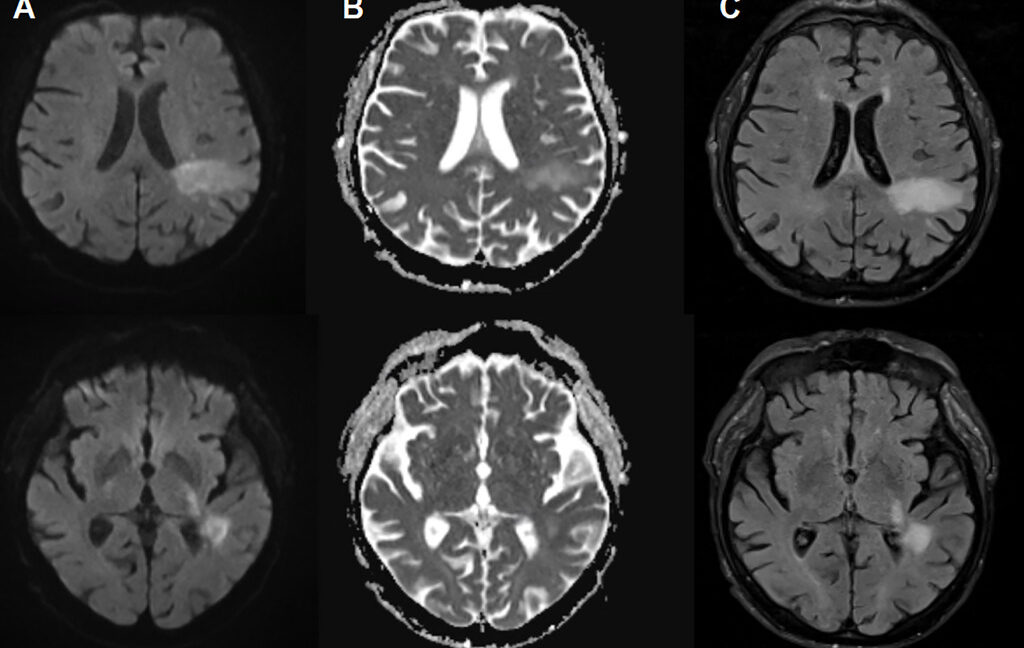
Foto: Annals of Internal Medicine: Clinical Cases, 2026
JC virus infects up to 90 percent of humanity, remaining dormant in cells throughout life, but when activated, it destroys the brain. Scientists recently discovered a new activation mechanism that could affect up to 10 percent of adults worldwide. Human polyomavirus 2, called JC virus or John Cunningham virus, spreads through the fecal-oral route, typically infects people in early childhood and remains asymptomatic throughout life. In rare cases, the virus mutates into a dangerous form, causing progressive multifocal leukoencephalopathy (PML) — a disease that destroys brain cells and damages myelin protecting nerves. Symptoms can mimic stroke or multiple sclerosis, leading to speech disorders, vision problems, motor dysfunction and seizures. PML was extremely rare until the 1980s, when it became common among HIV/AIDS patients. Before the introduction of HAART therapy in 1996, the disease was almost always fatal. Today, although survival is possible, patients often suffer permanent neurological damage. The discovery of a new mechanism for virus activation changes the perspective on understanding this threat.
There is a virus that you have probably never heard of, it infects up to 90 percent of the human population and lurks quietly in your cells throughout your life. But if it activates, it will destroy your brain. If that is not enough to worry you, scientists have just published research suggesting that there is a new way this virus can be activated — one that may affect up to 10 percent of adults worldwide. This discovery completely changes our current understanding of how and when this catastrophic infection occurs.
The virus we are talking about is human polyomavirus 2, commonly known as the JC virus or John Cunningham virus — named after the patient from whom the virus was first isolated in 1971. Since then, scientists have understood this disease better and better, but recent discoveries suggest that our current models of its spread and activation may be incomplete.
A secret passenger hidden in billions of people
The JC virus appears in the urine and feces of infected people and spreads through the fecal-oral route — that is, through contact with contaminated surfaces, water, or food. Scientists believe that most people contract this virus in early childhood, and blood tests suggest that 50 to 90 percent of adults have had contact with it at some point in their lives. This is a staggering number, which means that if you are an adult, the chance that you carry this virus is at least one in two.
Read also
Scientists hypothesize that the initial site of infection is the tonsils or perhaps the digestive tract. However, regardless of where the first contact with the virus occurs, this initial infection is completely asymptomatic. At this point, a person becomes infected with what scientists call the archetypal JC virus — a variant of the virus that quietly establishes a persistent, but completely unnoticed lifelong infection. For decades you can carry this virus and never know it, because your immune system keeps it in check.
For the vast majority of people, JC virus infection remains completely asymptomatic. But for a few unfortunate people, the virus seemingly awakens, rearranges its genetic material, and metamorphoses into a brain-destroying nightmare, causing a disease called progressive multifocal leukoencephalopathy or PML. This transition from an asymptomatic virus to a brain-destroying virus is one of the most sinister transformations in human pathology.
When the virus wakes from dormancy
In PML, a new variant of the virus — called the PML-type virus — actively enters the brain and destroys specific brain cells, including cells that create the insulating myelin sheaths that protect nerve cells. This leads to extensive demyelination, that is, damage to or loss of the myelin sheath surrounding neuronal axons, which in turn causes dysfunction and death of nerve cells. In medical images, PML appears as characteristic changes in the brain. These changes visible in images, combined with test results showing JC virus DNA in cerebrospinal fluid, are how PML is diagnosed.
For patients experiencing PML, symptoms can mimic anything — from stroke to multiple sclerosis, causing problems such as speech disorders, vision defects, motor dysfunction, and seizures. A patient may wake up with partial paralysis, lose sight or the ability to speak. The progression is usually rapid and relentless, and without treatment, the disease usually leads to death within months to a year.
PML was first identified in 1958 in a cancer patient, but for decades was considered an extremely rare disease. Everything changed in the 1980s when it began appearing in patients with HIV/AIDS. In fact, PML became an AIDS-defining illness, with 2–5 percent of HIV-infected patients developing it in the early stages of the epidemic. At that time, the disease was almost always fatal — patients diagnosed with PML had only a few months to live ahead of them.
A breakthrough that changed the trajectory of the disease
Everything changed with the introduction in 1996 of highly active antiretroviral therapy (HAART). This revolutionary combination of drugs could restore immune function in AIDS patients, allowing their bodies to control the JC virus. The number of PML cases dropped dramatically, and the disease ceased to be a death sentence. However, surviving patients often suffer from significant permanent neurological damage — persistent problems with movement, vision, or speech.
For many years, scientists thought they understood the mechanism of JC virus activation. They believed that the virus activates almost exclusively in people with profound immunosuppression — that is, in those who have a weakened immune system due to HIV/AIDS, taking immunosuppressive drugs after transplantation, or very rarely, in patients with other immune conditions. This explained why PML was so rare — most people have a strong enough immune system to keep the virus in check.
But the latest research is changing this narrative. Scientists have discovered that there may be a completely new mechanism of JC virus activation, one that does not require profound immunosuppression. This discovery suggests that the risk of PML may be much broader than previously thought, and that the virus may activate in people with a normally functioning immune system.
A new activation mechanism changes the game
The details of this new activation mechanism are still being clarified, but research suggests that it may affect up to 10 percent of adults worldwide. This would be staggering — if 10 percent of humanity has potentially increased risk of JC virus activation, it means that potentially hundreds of millions of people may be at risk. Of course, the fact that someone has this potential activation mechanism does not automatically mean they will develop PML — additional factors are still needed for the disease to develop.
Scientists are working to understand what these additional factors are. They could be specific genetic mutations, specific health conditions, exposure to specific environmental stimuli, or a combination of these factors. This is a complicated puzzle, and each new piece that scientists discover brings us closer to fully understanding why the virus activates in some people and remains dormant in others throughout their lives.
One thing is clear: this discovery has serious implications for public health. If significantly more people have potentially increased risk of JC virus activation, it means that PML may be more common than previously thought. This could mean the need to train doctors in recognizing the symptoms of PML, developing better diagnostic tests, and accelerating work on potential therapies.
Implications for patients and the healthcare system
For patients, this discovery can be both worrying and potentially empowering. Worrying, because it suggests that the risk of PML may be broader than previously thought. But potentially empowering, because greater awareness of this variant may lead to earlier diagnosis and potentially better outcomes. Patients who are aware of PML symptoms — vision disturbances, speech disturbances, muscle weakness — may seek medical attention more quickly if they experience these symptoms.
For the healthcare system, the discovery poses a challenge. Doctors will need to be more vigilant in looking for PML, especially in patients who may not fit the traditional profile — that is, people with profound immunosuppression. This may mean more aggressive testing in patients with atypical neurological symptoms, especially if traditional tests do not reveal a cause.
There is also potential for new therapeutic approaches. If scientists can better understand the new mechanism of JC virus activation, they may be able to develop interventions that prevent activation or inhibit the virus before it causes significant brain damage. These could be new drugs, immunotherapies, or combinations of existing approaches.
The significance of this discovery for future research
This discovery highlights the enormous gap in our knowledge about viruses that have coevolved with humanity for thousands of years. The JC virus is a perfect example of a virus that remains asymptomatic most of the time, but which can become catastrophically dangerous under certain conditions. Scientists have been studying the JC virus for decades, but this discovery of a new activation mechanism shows that we still have much to learn.
Research on the JC virus may also have broader implications for our understanding of other viruses. Many viruses, such as cytomegalovirus (CMV) or Epstein-Barr virus (EBV), can remain asymptomatic for long periods and then activate under certain conditions. If scientists can better understand the mechanisms of JC virus activation, this may lead to better understanding of the activation of these other viruses.
It is also worth noting that this discovery shows how important funding for basic research is. The scientists who made this discovery were probably not specifically looking for a new mechanism of JC virus activation — instead, they were engaged in long-term research on virus biology, and this discovery emerged from their work. This is a classic example of how basic research can lead to discoveries that have practical implications for public health.
Clinical perspective and future directions
While the discovery of a new mechanism of JC virus activation is significant, it is also important to maintain perspective. PML remains a rare disease — even if 10 percent of adults have potentially increased risk, it does not mean that all of them will become ill. Most people with this potential activation mechanism will never experience PML symptoms. However, for the few percent who do experience it, the disease can be catastrophic.
Clinicians are now working to develop better monitoring and intervention strategies for patients at higher risk. This may include regular testing for JC virus DNA in cerebrospinal fluid, monitoring for cognitive or neurological changes, and potentially prophylactic therapies for those identified as being at high risk. However, many of these approaches are still experimental, and further research is needed to determine which strategies are most effective.
Moreover, this discovery highlights the importance of personalized medicine. Not all people with JC virus will have this new activation mechanism, and not all people with this mechanism will have the same risk. Understanding the genetic and environmental factors that affect the risk of PML will be key to developing personalized approaches to prevention and treatment.
Ultimately, this discovery changes our perception of the JC virus — from a quiet, asymptomatic virus that activates only in those with profound immunosuppression, to a virus that potentially can activate in a much larger population through a new, not fully understood mechanism. This discovery is a reminder that the microorganisms we share with humanity can be far more complex and unpredictable than we thought, and that the science of infectious diseases is full of surprises.
More from Tech
Related Articles

After court loss, RFK Jr. gives himself more power over CDC vaccine panel
Apr 6
Steven Spielberg Still Wants to Make a Horror Film ‘Someday’
Apr 6New Jersey has no right to ban Kalshi's prediction market, US appeals court rules
Apr 6